SLIDE 1
Cp*Ru(PN) CATALYSTS WITH A NH/METAL BIFUNCTION R R Ru Ru H - - PowerPoint PPT Presentation
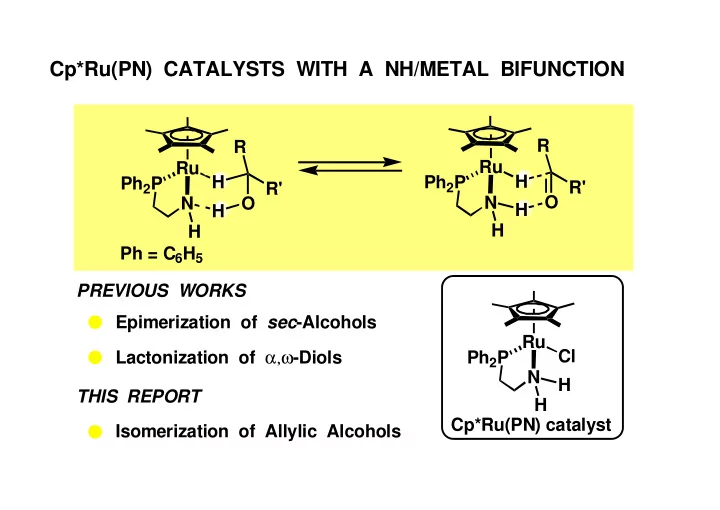
Cp*Ru(PN) CATALYSTS WITH A NH/METAL BIFUNCTION R R Ru Ru H - - PowerPoint PPT Presentation
Cp*Ru(PN) CATALYSTS WITH A NH/METAL BIFUNCTION R R Ru Ru H H Ph 2 P Ph 2 P R' R' O N N O H H H H Ph = C 6 H 5 PREVIOUS WORKS Epimerization of sec -Alcohols Ru Lactonization of , -Diols Cl Ph 2 P N H THIS REPORT H
SLIDE 2
SLIDE 3
Conditions; toluene, 30 °C, 1 h, alcohol:Cp*RuCl[Ph2P(CH2)2NH2]:KOt-Bu = 100:1:1
SCOPE AND LIMITATION
Ph OH R2 R3 R1 OH >99% OH >99% OH >99% HO >99% OH OH >99% OH X X N R1 = CH3, R2,R3 = H R2 = CH3, R1,R3 = H R1,R2 = CH3, R3 = H R1,R2 = ( CH2 )3 , R3 = H R1,R2 = ( CH2 )4 , R3 = H >99% conv. >99% >99% >99% >99% >99% R2,R3 = CH3, R1 = H R3 = CH3, R1,R2 = H R1,R3 = CH3, R2 = H R1,R2,R3 = CH3 67% 37% 23% X = O >99% = S >99% X = O >99% = S >99%
SLIDE 4
Ph OH O Ph O O O Ph O
MECHANISTIC CONSIDERATION -ENONE EXCHANGE REACTION-
Cp*RuCl[Ph2P(CH2)2NH2] KOt-Bu C6D6, 0.5 M, 30 °C, 1 h >99% conv. alcohol:enone:Cp*Ru:KOt-Bu = 100:100:1:1 24 % 76 % + + Intramolecular Intermolecular
SLIDE 5
R' O
A POSSIBLE MECHANISM
Ru Ph2P N H H Ru Ph2P Cp*RuCl[Ph2P(CH2)2NH2] + KOt-Bu R' OH R R R' O R + H H H NH H dehydrogenation reduction
SLIDE 6
EPIMERIZATION OF CHIRAL NON-RACEMIC sec-ALCOHOLS
Ph OH toluene Cp*RuCl[L(CH2)2NH2] Ph OH 99% ee L temp, °C time, h ee, % TOF, h-1 50 18 >99 21 PPh2 30 0.5 <1 479 alcohol:Cp*Ru:KOt-Bu = 100:1:1 KOt-Bu Tetrahedron Lett. 2003, 44, 7520 N(CH3)2
SLIDE 7
ISOTOPE LABELING EXPERIMENT
alcohol:Cp*Ru:KOt-Bu = 100:1:1
adetermined by 2H NMR (toluene-d8 was used as an internal standard)
OD2 30 °C Cp*RuCl[Ph2P(CH2)2NH2] OD2 D1 D1 toluene KOt-Bu ratio of deuteration, atom%Da D1 D2 time, min alcohol (CH3)2CDOH >99 30 68 32 60 51 49 (CH3)2CHOD 30 60 98 13 85 32 66
SLIDE 8
ISOTOPE LABELING EXPERIMENT
OH D OD H Cp*RuCl[Ph2P(CH2)2NH2] KOt-Bu C6D6 Ru Ph2P N D H H D O H Ru Ph2P N H Ru Ph2P N H D H
SLIDE 9
ISOTOPE LABELING EXPERIMENT
Ph OH D Ph O Cp*RuCl[Ph2P(CH2)2NH2] KOt-Bu Ru Ph2P N D H Ph O D H toluene, 30 °C TOF 471 h-1
>99% D >99% D <1% D
H
SLIDE 10
STEREOSPECIFICITY
Cp*RuCl[Ph2P(CH2)2NH2] KOt-Bu Ru Ph2P N H H O toluene, 30 °C, 1 h OH * O * (+)-(Z) 25% ee >99% yield 0% ee Ru Ph2P H NH2 H
SLIDE 11
OH O
ENANTIOSELECTIVE ISOMERIZATION
Cp*RuCl(cod) chiral ligand toluene 30 °C, 25 h * alcohol:Cp*Ru:ligand:KOt-Bu = 20:1:1:1 chiral ligand ( CH2 )3 Bn Ph t-Bu i-Pr yield, %a ee, %b 83 87 >99 >99 94 62 (S) 55 (S) 23 (S) 22 (S)
a determined by 1H NMR. b determined by GLC.
Ph2P NHR2 R1 chiral ligand KOt-Bu R1 R2 H H H H 44 (R) substrate Z E Z E Z E Z E Z E ( CH2 )3 Bn H Ph H i-Pr H t-Bu H >99 >99 >99 >99 n.d. 66 (R) 66 (R) 33 (R) 39 (R) 28 (S)
SLIDE 12
OH I O (2) Cp*RuCl[Ph2P(CH2)2NH2] toluene, 0.5 M, 30 °C, 1 h S/C = 100 O O KOt-Bu
A NOVEL EFFICIENT ROUTE TO DL–MUSCONE
(1) t-BuLi (2 equiv.) n-C5H12–Et2O –78°C 78% yield >99% conv. 76% yield Grubbs Cat.2nd G. (±) ClCH2CH2Cl, 65 °C 0.001 M, S/C = 10 >99% conv. 68% yield H2 / Pd-C EtOH, rt >99% conv. 90% yield (±) (±)–Muscone ( )8 ( )8 O ( )8
SLIDE 13
ASYMMETRIC SYNTHESIS OF THE MUSCONE PRECURSOR
alcohol:Ru:KOt-Bu = 20:1:1 toluene 30 °C Cp*RuCl(PN*) OH (±) O * NHR2 Ph2P R1 chiral ligand; PN* substrate Z E yield, % ee, %a E E 81 95 Bn i-Pr 83 74 (S) 86 KOt-Bu R1 R2 27 (R) 64 (R) 1 (R) H H ( )8 ( )8 chiral ligand
aDetermined by HPLC after conversion to Muscone
(CH2)3 (CH2)3
SLIDE 14