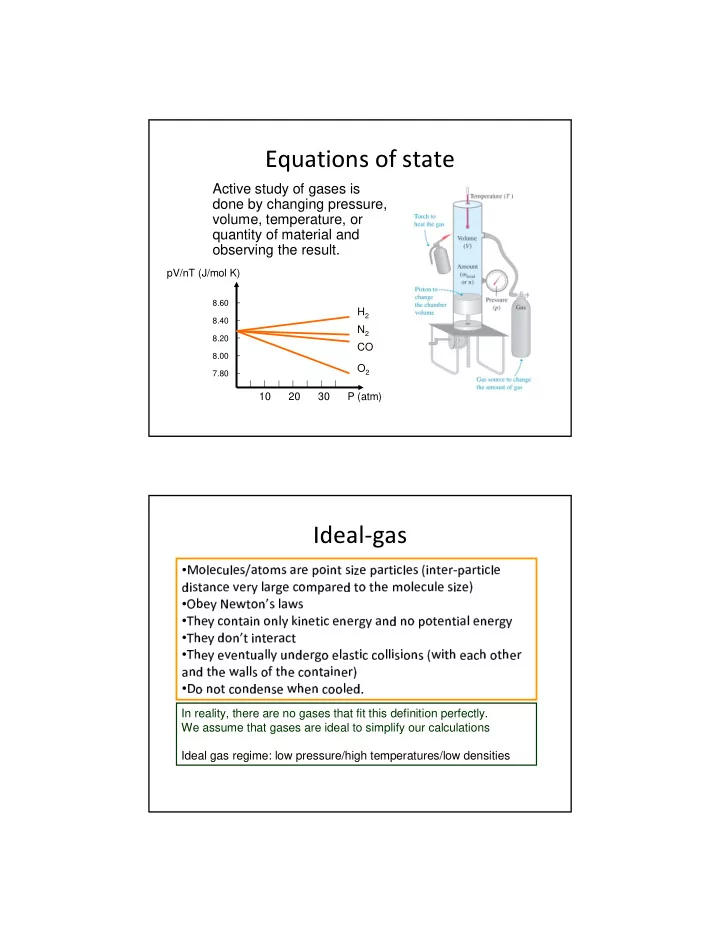
Equations of state
Active study of gases is done by changing pressure, volume, temperature, or quantity of material and
- bserving the result.
8.60 8.40 8.20 8.00 7.80
10 20 30 P (atm) pV/nT (J/mol K) H2 N2 CO O2
Ideal-gas
- ✁
- ✘
- ✢
- ✢
- ✢
- ✥
In reality, there are no gases that fit this definition perfectly. We assume that gases are ideal to simplify our calculations Ideal gas regime: low pressure/high temperatures/low densities