SLIDE 1
HYDROGEN TRANSFER REACTIONS OF ALCOHOLS Racemization of sec - - PowerPoint PPT Presentation
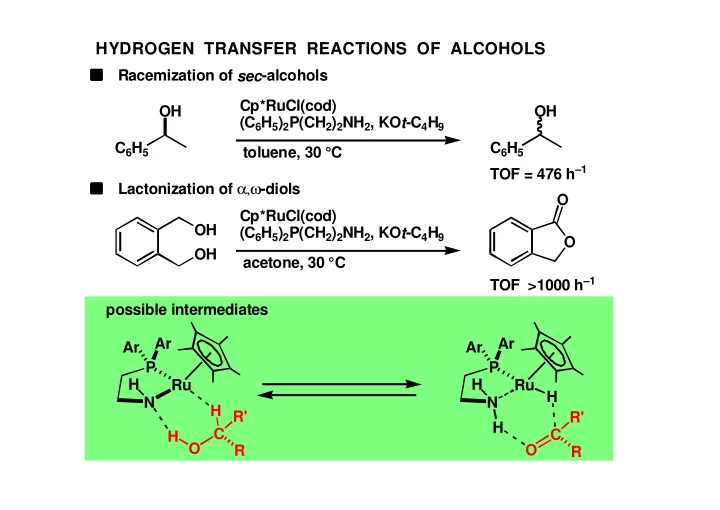
HYDROGEN TRANSFER REACTIONS OF ALCOHOLS Racemization of sec - - PowerPoint PPT Presentation
HYDROGEN TRANSFER REACTIONS OF ALCOHOLS Racemization of sec -alcohols Cp*RuCl(cod) OH OH (C 6 H 5 ) 2 P(CH 2 ) 2 NH 2 , KO t -C 4 H 9 C 6 H 5 C 6 H 5 toluene, 30 C TOF = 476 h 1 Lactonization of , -diols O Cp*RuCl(cod) OH (C 6 H
SLIDE 2
SLIDE 3
O R1N O R2 HP(C6H5)2
S
SYNTHESIS OF OPTICALLY ACTIVE P–N LIGANDS
S
H2N P(C6H5)2 R NH P(C6H5)2 R = CH3 = CH(CH3)2 = CH2CH(CH3)2 = C(CH3)3 = C6H5 = CH2C6H5 83% yield 89 85 66 70 70
S
55% yield H +
日本化学会第81春季年会,3G414
SLIDE 4
O HN O R2 HPR2 CF3SO3H
S S
H2N PR2 R2
SYNTHESIS OF OPTICALLY ACTIVE P–N LIGANDS
+ toluene reflux, 24 h Conditions; oxazolidinone:HPAr2:CF3SO3H = 1:2:3 K2CO3 aq
S
H2N PR2 C6H5H2C 69% yield R = p-CH3C6H4 3,5-(CH3)2C6H3 66% R1 R1
S
H2N P(C6H5)2 R2 C6H5 R2 = CH3 77% yield 54% C6H5 C6H11 <1%
SLIDE 5
R1HN PAr2 R2 R3 CH2Cl2 Ru PAr2 Cl N R1 R2 H R3 Ar H, Bn, H C6H5 C6H5 C6H5 C6H5 C6H5 C6H5 C6H5 C6H5 H, Bn, H H, Bn, H 92 93 90 C6H5 99 88 82 85 88 86 89 82 Cp*RuCl(isoprene) +
SYNTHESIS OF Cp*RuCl(P–N) COMPLEXES
H, i-Pr, H H, i-Bu, H H, CH3, H R1, R2, R3 H, C6H5, C6H5 p-CH3C6H4 3,5-(CH3)2C6H3 H, t-Bu, H H, C6H5, H
- (CH2)3-, H
yield, % H, H, H 60.9 59.6, 61.8 54.0 54.8 55.8 56.5 57.1 56.6 55.2 70.1
31P NMR
56.3 1:1
SLIDE 6
Selected bond lengths [Å] and angles [°] Ru(1)—N(1) Ru(1)—P(1) Ru(1)—Cl(1) N(1)–Ru(1)–P(1) N(1)–Ru(1)–Cl(1) Cl(1)–Ru(1)–P(1) 2.183(6) 2.302(2) 2.495(2) 79.7(2) 83.7(2) 93.54(9)
SYNTHESIS OF Cp*RuCl(P–N) COMPLEX
Selected bond lengths [Å] and angles [°]a Ru(1)—N(1) Ru(1)—P(1) Ru(1)—Cl(1) N(1)–Ru(1)–P(1) N(1)–Ru(1)–Cl(1) Cl(1)–Ru(1)–P(1) 2.1915(3) 2.2885(10) 2.4860(10) 81.25(7) 79.58(9) 91.49(5) P1 (#1), R1 0.029, wR2 0.077 P212121 (#19), R1 0.078, wR2 0.202
Ru1 Cl1 N1 P1 H2 H1 Ru1 N1 P1 Cl1 H1 H2
H2N PAr2 Bn Ar = p-CH3C6H4 Ar = 3,5-(CH3)2C6H3
R S S S
P–N
aaverage of two indipendent molecules
SLIDE 7
N P(C6H5)2 Cp*RuCl(isoprene) +
SYNTHESIS OF Cp*RuCl(P–N) COMPLEX
CH2Cl2 H P21/c (#14) R 0.063 RW 0.062 Ru1 Cl1 P1 N1 Ru P(C6H5)2 Cl N H H r.t. –90 °C
50 1.0 0.9 1.0 1.0 S S
SLIDE 8
H2N P(C6H5)2 Cp*RuCl(isoprene) +
SYNTHESIS OF Cp*RuCl(P–N) COMPLEX
CH2Cl2 Ru P(C6H5)2 Cl H2N P21 (#4) R1 0.028 wR2 0.083 N1 P1 Ru1 Cl1 Cl2 P2 Ru2 N2 Ru P(C6H5)2 Cl H2N +
S S R
SLIDE 9
31P NMR
Ru P(C6H5)2 Cl H2N
EPIMERIZATION OF RUTHENIUM CENTRAL CHIRALITY
r.t. –90 °C –50 °C
56 57 58 59 1.0 10.0 1.0 13.0
Ru P(C6H5)2 Cl H2N
SLIDE 10
REACTION OF Cp*RuCl(P–N) COMPLEX
Ru P(C6H5)2 Cl H2N CO (1 atm) Ru P(C6H5)2 CO HN 1.64 ppm, d JP-H= 1.7 Hz, 15H, C5(CH3)5 methylene bridge 2.05-2.15 ppm, m, 1H 2.60-2.71, m, 1H 2.87-3.01, m, 1H 3.40-3.51, m, 1H 6.63, br, 1H, NH 7.22-7.62, m, 10H, aromatics
1H NMR(CD2Cl2) 13C NMR
10.0 ppm, s, C5(CH3)5 32.0, d, JCP = 27.5 Hz, PCH2 47.2, d, JCP = 5.4 Hz, NCH2 96.1, d, JCP = 1.9 Hz, C5(CH3)5 C6H5 128.7 ppm, d, JCP = 10.3 Hz 129.2, d, JCP = 10.3 Hz 130.2, d, JCP = 2.7 Hz 131.5, d, JCP = 2.7 Hz 132.8, d, JCP = 10.7 Hz 133.5, d, JCP = 49.6 Hz 134.4, d, JCP = 12.2 Hz 135.2, d, JCP = 42.7 Hz FT-IR 1933 cm-1, νCO 98% yield 64.5 ppm, s
31P NMR
Ru P(C6H5)2 HN KOH CO 205.6 ppm, d JP-H = 16.4 Hz, CO
SLIDE 11
REACTION OF Cp*RuH(P–N) COMPLEX WITH ACETONE
Ru P(C6H5)2 H H2N 96% yield
- 10.6 ppm, d, JPH = 40.3 Hz, 1H, Ru-H
1.9, d, JPH = 0.9 Hz, 15H, C5(CH3)5 3.0, m, 2H, methylene bridge 4.1, m, 2H, methylene bridge 7.4-8.2, m, 10H, aromatics
1H NMR(THF-d8)
79.4 ppm, s
31P NMR
Ru P(C6H5)2 Cl H2N KOH 2-propanol
SLIDE 12
EARLY EXAMPLES FOR Ru–CATALYZED LACTONIZATION
Ru2Cl4((-)diop)3 PhCH=CHCOCH3, 2 equiv toluene, reflux OH OH n n O O Ishii, Osakada, Ikariya, Saburi, Yoshikawa, Chem. Lett., 1982, 1179. up to 12% ee OH OH [RuCl((S)-binap)(benzene)]Cl PhCH=CHCOCH3 (2 equiv) toluene, SDS, 60 °C, 44 h O O 89% yield 11% ee Nozaki, Yoshida, Takaya, J. Organomet. Chem., 1994, 473, 253. OH OH Ru(salen) hυ air, CHCl3 r.t., 2-3 d O up to 67% ee O n n O OH n PDC, MS4A CH2Cl2 Shimizu, Nakata, Katsuki, Chem. Lett., 2002, 1080. S/C = 25 S/C = 100 S/C = 50
SLIDE 13
OH OH Ar Ph Ph Ph Ph Ph Ph Ph O O
S R
ENANTIOSELECTIVE LACTONIZATION FROM meso-DIOLs
Cp*RuCl(P–N) KOt-Bu acetone 30 °C, 1 h p-CH3C6H4 3,5-(CH3)2C6H3 yield, % ee, % diol:Ru:chiral ligand:KOt-Bu = 100:1:1:1 >99 >99 >99 >99 >99 >99 45 45 39 41 46 10 >99 >99 44 41 >99 43 R1, R2, R3 H, Bn, H H, Bn, H H, Bn, H H, t-Bu, H H, Ph, H
- (CH2)3-, H
H, i-Pr, H H, i-Bu, H H, Me, H Ru PAr2 Cl N R1 R2 H R3 Cp*RuCl(P–N)
SLIDE 14
KINETIC RESOLUTION OF rac-1,4-BUTANDIOL
diol:acetone:Ru:chiral ligand:KOt-Bu = 100:100:1:1:1 Ar Ar OH OH OH OH Ar Ar OH OH Ar Ar O Ar Ar O + Cp*RuCl(P–N) KOt-Bu acetone toluene 30 °C, 8 h + 50:50 Ar = C6H5 (2R, 3S) (2S, 3R) kf/ks = 33.0 26% conv. 33% ee 92% ee Ru P(C6H5)2 Cl H2N C6H5 Cp*RuCl(P–N)
SLIDE 15