SLIDE 1
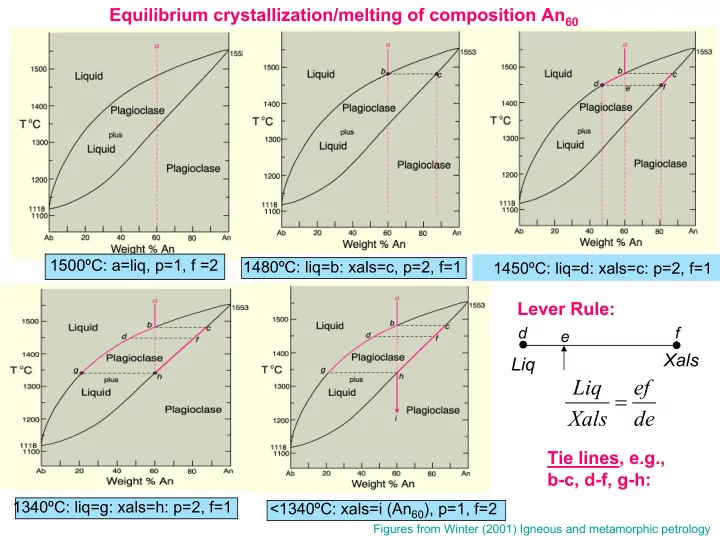
Fractional crystallization/melting of composition An60
With fractional crystallization of bulk compn a, first crystals have compn c. Crystals are removed from system as soon as they form so are not present to react with liquid. The liquid will fractionate to pure albite compn and the total range of plagioclase crystals will be from c to pure Ab. With fractional melting of compn i, the first liquid to form will have compn g. Each fraction of melt produced is removed from the system so the solid assemblage changes compn from h to pure An and the liquid fractions range from g to pure
- An. Mean compn of liquid is a