Madhav Marathe Dept. of Computer Science & Network Dynamics and Simulation Science Laboratory Virginia Bioinformatics Institute Virginia Tech NDSSL TR09074
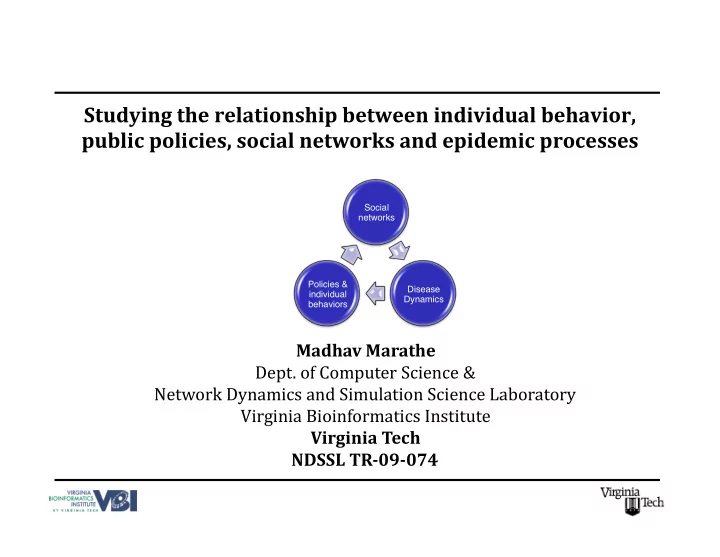
Studying the relationship between individual behavior, public policies, social networks and epidemic processes
Social networks Disease Dynamics Policies & individual behaviors