SLIDE 1
Carey Chapter 2 Hydrocarbon Frameworks Alkanes Hydrocarbons - - PowerPoint PPT Presentation
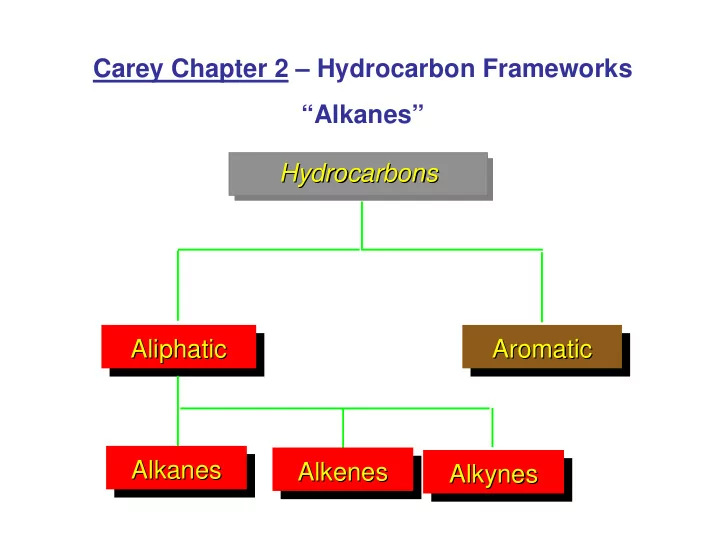
Carey Chapter 2 Hydrocarbon Frameworks Alkanes Hydrocarbons - - PowerPoint PPT Presentation
Carey Chapter 2 Hydrocarbon Frameworks Alkanes Hydrocarbons Hydrocarbons Hydrocarbons Aliphatic Aromatic Aliphatic Aromatic Aliphatic Aromatic Alkanes Alkanes Alkenes Alkenes Alkynes Alkynes Alkanes Alkenes Alkynes
SLIDE 2
SLIDE 3
2.2-2.3 Chemical Bonding – Two Possibilities Figure 2.5
SLIDE 4
2.4 Molecular orbitals by combining two atomic orbitals Figure 2.6
SLIDE 5
2.5 Introduction to Alkanes – Methane, Ethane, Propane
Figure 2.7
CH4 CH3CH3 CH3CH2CH3 b.p. -160 oC
- 89 oC
- 42 oC
SLIDE 6
2.6 sp3 Hybridization and bonding in Methane
Figure 2.9
SLIDE 7
2.6 sp3 Hybridization and bonding in Methane
Figure 2.10
SLIDE 8
2.7 sp3 Hybridization and bonding in Ethane
Figure 2.11
SLIDE 9
2.8 Isomeric alkanes – the Butanes Structural Isomers C5H12 neopentane C4H10 isobutane C5H12 n-pentane C5H12 isopentane C4H10 n-butane 2.9-2.10 Higher alkanes – the C5H12 isomers
SLIDE 10
2.10 Higher alkanes – diversity
SLIDE 11
Careful with drawing chains!
CH3CHCH2CH3 CH3 CH3CHCH2CH3 CH3 CH3CH2CHCH3 CH3 CH3CH2CHCH3 CH3 CH2CH2CH3 CH3 CH3
All the same compound
SLIDE 12
2.11-2.12 Alkane nomenclature Need to know up to C-12
SLIDE 13
IUPAC Rules:
- Find the longest continuous carbon chain
- Identify substituent groups attached to the chain
- Number the chain so as to keep numbers small
- Write the name in the following format:
Numerical location - [substituent(s)][parent alkane] e.g. 2,3-dimethylheptane 2.11-2.12 Alkane nomenclature
SLIDE 14
2.12 IUPAC Rules and how to apply them
Hexane (IUPAC); n-hexane (common) Longest chain - hexane substituent - methyl position on chain - 2 2-methylhexane not 5-methylhexane 3,4-dimethylheptane
SLIDE 15
2.13 Alkyl groups Replace -ane ending with -yl
H C H C C C H C C C C C
primary (1o) secondary (2o) tertiary (3o) propyl group isopropyl group t-Butyl group 1-methylethyl 1,1-dimethylethyl
CH3 C CH3 H3C C H H H C H H C H H CH3CH2CH2 CH CH3 CH3 (CH3)2CH (CH3)3C
SLIDE 16
2.14 Highly branched alkanes 4-ethyloctane 4-ethyl-3-methyloctane 4-ethyl-3,5-dimethyloctane
SLIDE 17
2.15 Cycloalkanes
1,1,3-trimethylcyclohexane C(CH3)3 (1,1-dimethylethyl)cycloheptane 2-ethyl-1,1- dimethylcyclopentane (notice the “di” is not involved in the alphabetization)
SLIDE 18
2.16 Sources of alkanes and cycloalkanes Figure 2.12
SLIDE 19
2.17 Physical properties Figure 2.15
SLIDE 20
2.17 Physical properties – branched alkanes Figure 2.16
SLIDE 21
2.18 Chemical properties of Alkanes Alkane properties:
- Generally very insoluble in water (“greasy” or “oily”)
- Individual molecules interact via van der Waals forces
- These intermolecular forces decrease with branching
- Alkanes may be combusted in oxygen:
e.g. CH4 + 2O2 CO2 + 2H2O ∆H = - 213 kcal i.e combustion of hydrocarbons releases energy
SLIDE 22
2.18 Heats of combustion – Figure 2.17
SLIDE 23
2.19 Oxidation-Reduction in Organic Chemistry
SLIDE 24
C C H H H H
2.20 sp2 Hybridization in ethylene Figure 2.18
SLIDE 25
2.20 sp2 Hybridization in ethylene Figure 2.19
SLIDE 26
2.20 sp2 Hybridization in ethylene Figure 2.20
SLIDE 27
2.21 sp Hybridization in ethylene Figure 2.21
SLIDE 28
2.21 sp Hybridization in acetylene Figure 2.22
SLIDE 29