Matter and Energy Notes.notebook 1
Matter and Energy Chapter 1: Introduction to Matter
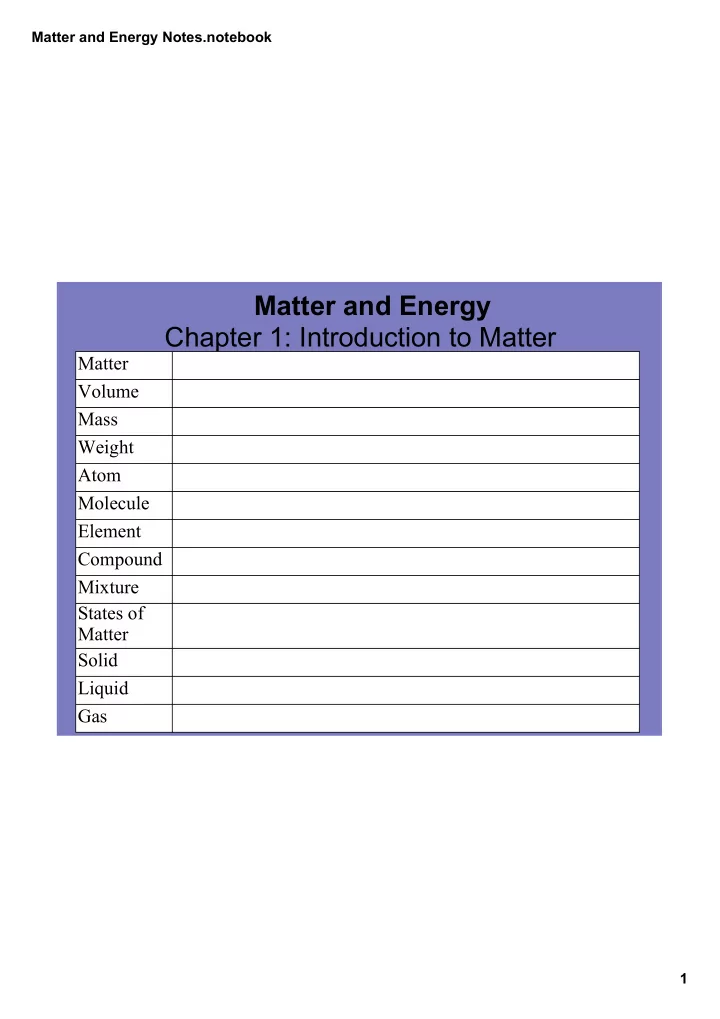
Matter Volume Mass Weight Atom Molecule Element Compound Mixture States of Matter Solid Liquid Gas
Matter and Energy Chapter 1: Introduction to Matter Matter Volume - - PDF document
Matter and Energy Notes.notebook Matter and Energy Chapter 1: Introduction to Matter Matter Volume Mass Weight Atom Molecule Element Compound Mixture States of Matter Solid Liquid Gas 1 Matter and Energy Notes.notebook Matter and
Matter and Energy Notes.notebook 1
Matter and Energy Chapter 1: Introduction to Matter
Matter Volume Mass Weight Atom Molecule Element Compound Mixture States of Matter Solid Liquid Gas
Matter and Energy Notes.notebook 2
Matter and Energy Chapter 1: Introduction to Matter
Matter and Energy Notes.notebook 3
Matter and Energy Chapter 1: Introduction to Matter
Matter and Energy Notes.notebook 4
Matter and Energy Chapter 1: Introduction to Matter
Matter and Energy Notes.notebook 5
Matter and Energy Chapter 1: Introduction to Matter
Matter and Energy Notes.notebook 6
Matter and Energy Chapter 1: Introduction to Matter
Matter and Energy Notes.notebook 7
Matter and Energy Chapter 1: Introduction to Matter
Matter and Energy Notes.notebook 8
Matter and Energy Chapter 1: Introduction to Matter
Matter and Energy Notes.notebook 9
Matter and Energy Chapter 1: Introduction to Matter
Matter and Energy Notes.notebook 10
Matter and Energy Chapter 1: Introduction to Matter
Matter and Energy Notes.notebook 11
Matter and Energy Chapter 1: Introduction to Matter
Matter and Energy Notes.notebook 12
Matter and Energy Notes.notebook 13
Matter and Energy Notes.notebook 14
Matter and Energy Chapter 1: Introduction to Matter
Matter and Energy Notes.notebook 15
Matter and Energy Chapter 1: Introduction to Matter
SHOW ALL WORK
Matter and Energy Notes.notebook 16
Matter and Energy Chapter 1: Introduction to Matter
Matter and Energy Notes.notebook 17
Matter and Energy Notes.notebook 18
Matter and Energy Chapter 1: Introduction to Matter Step 1: Put water into a graduated cylinder and write down the volume: Step 2: Slow and carefully slide the object you want to measure into the graduated cylinder. Step 3: Read the new volume on the graduated cylinder. Step 4: Subtract the volume of the water before from the volume of the water after you added the object.
Matter and Energy Notes.notebook 19
Matter and Energy Notes.notebook 20
Matter and Energy Notes.notebook 21
Matter and Energy Notes.notebook 22
Matter and Energy Notes.notebook 23
Matter and Energy Chapter 1: Introduction to Matter
show work
Matter and Energy Notes.notebook 24
Matter and Energy Chapter 1: Introduction to Matter
show work
Mass: 2grams
Matter and Energy Notes.notebook 25
Matter and Energy Chapter 1: Introduction to Matter
show work
Matter and Energy Notes.notebook 26
Matter and Energy Chapter 1: Introduction to Matter
show work
Matter and Energy Notes.notebook 27
Matter and Energy Notes.notebook 28
Matter and Energy Notes.notebook 29
Matter and Energy Notes.notebook 30
Matter and Energy Notes.notebook 31
A cork and a pebble are each placed in 100mL of water, and each displaces 10mL of water. The cork floats, but the pebble sinks. How do the volumes of the cork and the pebble compare?
b.The cork’s volume is greater c.The pebbles volume is greater d.The volumes cannot be compared using this method
Matter and Energy Notes.notebook 32
Matter and Energy Notes.notebook 33
Matter and Energy Notes.notebook 34
Matter and Energy Notes.notebook 35
called:
in the nucleus.
in the nucleus.
fly around nucleus.
Matter and Energy Notes.notebook 36
Matter and Energy Notes.notebook 37
Matter and Energy Notes.notebook 38
Matter and Energy Notes.notebook 39
Matter and Energy Notes.notebook 40
a._____ atom(s) of ______________________ _____ atom(s) of _______________________ b.Total number of atoms = ____________
Matter and Energy Notes.notebook 41
a.1 atom(s) of Carbon 2 atom(s) of Oxygen b.Total number of atoms = 3
Matter and Energy Notes.notebook 42
a._____ atom(s) of ______________________ _____ atom(s) of _______________________ b.Total number of atoms = ____________
Matter and Energy Notes.notebook 43
1 atom(s) of Oxygen b.Total number of atoms = 2
Matter and Energy Notes.notebook 44
a._____ atom(s) of ______________________ _____ atom(s) of _______________________ _____ atom(s) of ______________________ b.Total number of atoms = ____________ ++++
Matter and Energy Notes.notebook 45
12 atom(s) of Hydrogen 6 atom(s) of Oxygen b.Total number of atoms = 24
Matter and Energy Notes.notebook 46
a._____ atom(s) of ______________
Matter and Energy Notes.notebook 47
a.3 atom(s) of Oxygen
3
Matter and Energy Notes.notebook 48
a._____ atom(s) of ______________________ _____ atom(s) of _______________________ _____ atom(s) of ______________________ b.Total number of atoms = ____________
Matter and Energy Notes.notebook 49
a.2 atom(s) of Hydrogen 1 atom(s) of Sulfur 4 atom(s) of Oxygen b.Total number of atoms = 7
Matter and Energy Notes.notebook 50
a._____ atom(s) of ______________________ _____ atom(s) of _______________________ b.Total number of atoms = ____________
Matter and Energy Notes.notebook 51
a.1 atom(s) of Hydrogen 1 atom(s) of Chlorine b.Total number of atoms = 2
Matter and Energy Notes.notebook 52
1.3: MATTER COMBINES TO FORM DIFFERENT SUBSTANCES
PURE SUBSTANCE: has only one
Ex: pure water - only water, no minerals or other chemicals
MIXTURES: more than one substance in the same place at the same time but not bonded
Matter and Energy Notes.notebook 53
Pure Substances are Atoms, Elements, and Compounds Examples: C = Carbon (element 1 atom) CO2 = Carbon Dioxide: Compound
(2 or more atoms bonded together)
Matter and Energy Notes.notebook 54
ATOMS determine the type of substance
Ex - a bar of pure gold, is made up of identical "gold" atoms
ELEMENTS: a type of pure substance that contains only one type of atoms
All the elements are organized on the periodic table on the last page of the chemical interactions text book
PURE SUBSTANCES ARE.........
Matter and Energy Notes.notebook 55
COMPOUND: substance that consists of 2 or more different types of atoms (elements) bonded together.
Matter and Energy Notes.notebook 56
Matter and Energy Notes.notebook 57
Matter and Energy Notes.notebook 58
together
separated physically
compound are the same throughout the compound
number of atoms is always the same
bonded
physically
based on substance.
Matter and Energy Notes.notebook 59
Mixtures Heterogenous vs. Homogenous
Can see the individual parts
Cannot see the
same throughout.
SALT WATER
Matter and Energy Notes.notebook 60
Matter and Energy Notes.notebook 61
determine the state of matter.....
arrangement of its molecules changes, the distance between molecules and the attraction between molecules changes.
Matter and Energy Notes.notebook 62
Matter and Energy Notes.notebook 63
SOLID: Fixed (doesn't change) volume Fixed shape Rigid: particles are close together and stay in one place.
Matter and Energy Notes.notebook 64
LIQUID: Fixed volume No fixed shaped: takes shape
Particles not fixed: particles are close together but are free to move around
Matter and Energy Notes.notebook 65
Matter and Energy Notes.notebook 66
Evaporation Condensation Freezing
Melting
increase in energy(heat) decrease in (energy) heat
Matter and Energy Notes.notebook 67
Matter and Energy Notes.notebook 68
When you increase the pressure on a gas, the volume decreases When you decrease the pressure on a gas, the volume increase.
(inverse proportion)
Matter and Energy Notes.notebook 69
Matter and Energy Notes.notebook 70
Matter and Energy Notes.notebook 71
As the temperature of a gas increases, the volume of the gas increases. As the temperature of the gas decreases, the volume of the gas decreases.
(direct proportion)
Matter and Energy Notes.notebook 72
Temperature and Volume:
Matter and Energy Notes.notebook 73